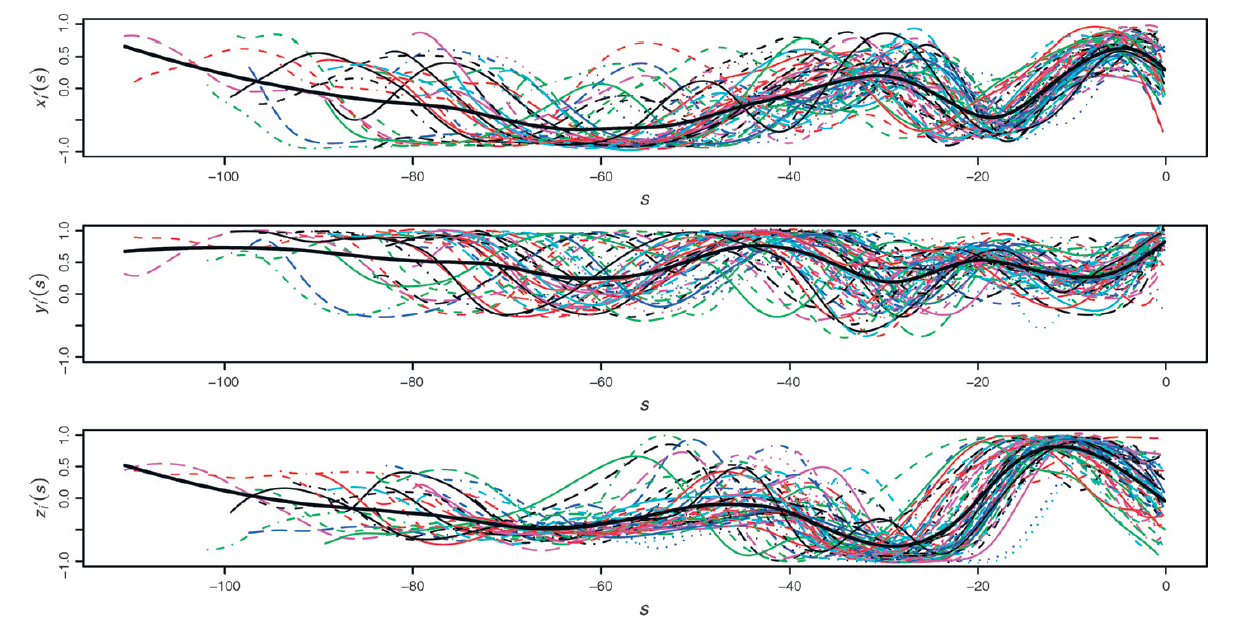
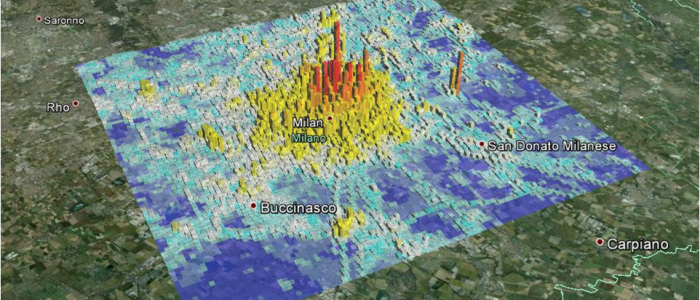
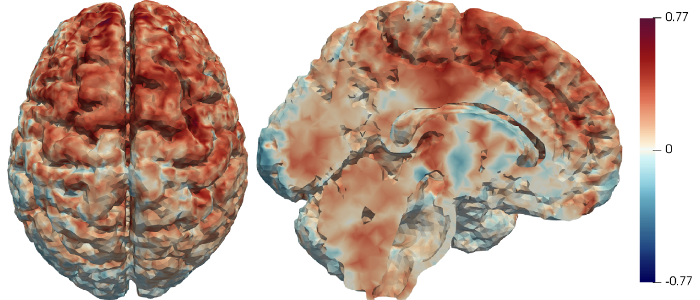
The research activity of the Statistics group at MOX is focused on the development of statistical models and methods for high dimensional and complex data, driven by industrial problems and problems arising in the life, environmental and social sciences.
Keywords:
Statistical Modelling of Complex and High Dimensional Data, Functional Data Analysis, Geostatistics, Nonparametrics, Penalized Regression, Compositional Data, Big data, Data Mining, Health Analytics.